Monoamine Oxidase Inhibitors for Neurological Disorders: A review
Abstract
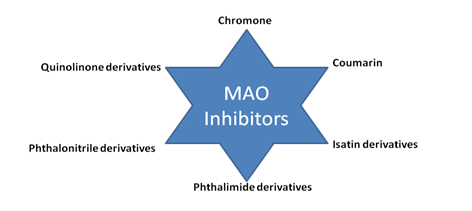
Neurological disorders are drawing much attention from scientists in recent times. These disorders are one of the most serious disorders mankind will have to fight against in the very near future. Monoamine oxidase is one target which is responsible for many neurological disorders. Scientists have studied different molecular entities as monoamine oxidase inhibitors. The MAO-A and -B inhibitors have been developed to increase the available amounts of monoamine neurotransmitters in the brains for the treatment of neurological disorders. In this article, selected classes of compounds like coumarins, chromones, isatins, phthalimides, phthalonitriles and quinolinones have been shown as potential inhibitors of human MAO-A and MAO-B.
Keywords
References
G.C. Griffith. Amine Oxidase Inhibitors: Their current place in the therapy of cardiovascular diseases. Circulation 1960, 22, 1156-1165.
C. Binda, P. Newton-Vinson, F. Hubálek, D.E. Edmondson, A. Mattevi. Structure of human monoamine oxidase B, a drug target for the treatment of neurological disorders. Nat. Struct. Biol. 2002, 9, 22–26.
K.F. Tipton, S. Boyce, J. O’Sullivan, G.P. Davey, J. Healy. Monoamine oxidases: certainties and uncertainties. Curr. Med. Chem. 2004, 11, 1965–1982.
D.E. Edmondson, A. Mattevi, C. Binda, M. Li, F. Hubálek. Structure and mechanism of monoamine oxidase. Curr. Med. Chem. 2004, 11, 1983–1993.
M. Reyes-Parada, A. Fierro, A.P. Iturriaga-Vasquez, B.K. Cassels. Monoamine oxidase inhibition in the light of new structural data. Curr. Enzyme Inhib. 2005, 1, 85–95.
S.E. Rigby, J. Basran, J.P. Combe, A.W. Mohsen, H. Toogood, A. van Thiel, M.J. Sutcliffe, D. Leys, A.W. Munro, N.S. Scrutton. Flavoenzyme catalysed oxidation of amines: roles for flavin and proteinbased radicals. Biochem. Soc. Trans. 2005, 33, 754–757.
R.H. Haung, R. Faulkner. The role of phospholipid in the multiple functional forms of brain monoamine oxidase. J. Biol. Chem. 1981, 256, 9211-9215.
A.W.J. Bach, N.C. Lan, D.J. Bruke, C.W. Abell, M.E. Bembenek, S.W. Kwan, P.H. Seeburg, J.C. Shih. Molecular-cloning of human monoamine oxidase-A and oxidase-B (MAO-A and MAO-B). FASEB J. 1988, 2, A1733-A1733.
K. Chen, H.â€F. Wu, J.C. Shih. The deduced amino acid sequences of human platelet and frontal cortex monoamine oxidase B are identical. J. Neurochem. 1993, 61, 187-190.
T. Okaecwe, A.J. Swanepoel, A. Petzer, J.J. Bergh, J.P. Petzer, Inhibition of monoamine oxidase by 8-phenoxymethylcaffeine derivatives. Bioorg. Med. Chem. 2012, 20, 4336–4347.
A.W.J. Bach, N.C. Lan, D.L. Johnson, C.W. Abell, M.E. Bembenek, S.W. Kwan, P.H. Seeburg, J.C. Shih. cDNA cloning of human liver monoamine oxidase A and B: molecular basis of differences in enzymatic properties. Proc. Natl. Acad. Sci. U.S.A. 1988, 85, 4934–4938.
J. Wouters. Structural Aspects of Monoamine Oxidase and its Reversible Inhibition. Curr. Med. Chem. 1998, 5, 137-162.
J. Grimsby, N.C. Lan, R. Neve, K. Chen, J.C. Shih. Tissue Distribution of Human Monoamine Oxidase A and B mRNA. J. Neurochem. 1990, 55, 1166-1169.
A.S. Kalgutkar, N. Castagnoli, B. Testa. Selective inhibitors of monoamine oxidase (MAO-A and MAO-B) as probes of its catalytic site and mechanism. Med. Res. Rev. 1995, 15, 325–388.
R.M. Geha, I. Rebrin, K. Chen, J.C. Shih. Substrate and inhibitor specificities for human monoamine oxidase A and B are influenced by a single amino acid. J. Biol. Chem. 2001, 276, 9877–9882.
P.H. Yu, B.A. Davis, A.A. Boulton. Neuroprotective effects of some monoamine oxidase-B inhibitors against DSP-4-induced noradrenaline depletion in the mouse hippocampus. J. Neurochem. 1994, 63, 1820-1828.
T. Thomas. Monoamine oxidase-B inhibitors in the treatment of Alzheimer’s disease. Neurobiol. Aging. 2000, 21, 343-348.
E.E. Billett. Monoamine Oxidase (MAO) in human peripheral tissues. NeuroToxicology. 2004, 25, 139–148.
S.-Y. Son, J. Ma, Y. Kondou, M. Yoshimura, E. Yamashita, T. Tsukihara. Structure of human monoamine oxidase A at 2.2-Å resolution: The control of opening the entry for substrates/inhibitors. Proc. Natl Acad. Sci. USA. 2008, 105, 5739–5744.
M.C. Anderson, F. Hasan, J.M. McCrodden, K.F. Tipton. Monoamine oxidase inhibitors and the cheese effect. Neurochem. Res. 1993, 18, 1145–1149.
C.I. Manley-King, J.J. Bergh, J.P. Petzer. Monoamine oxidase inhibition by C4-substituted phthalonitriles. Bioorg. Chem. 2012, 40, 114–124.
http://www.who.int/features/qa/55/en/ (Accessed on: 01 December 2013)
http://www.who.int/mental_health/neurology/chapter_2_neuro_disorders_public_h_
challenges.pdf (Accessed on: 01 December 2013)
http://www.who.int/mediacentre/news/releases/2007/pr04/en/ (Accessed on: 01 December 2013)
K.C. Teo, S.-L. Ho. Monoamine oxidase-B (MAO-B) inhibitors: implications for disease-modification in Parkinson’s disease. Translat. Neurodegen. 2013, 2, 19-28.
E.C. Coelho, P.A. Netz, V.P. do Canto, A.P. Cunha, C. Follmer. Beyond topoisomerase inhibition: antitumor 1,4-naphthoquinones as potential inhibitors of human monoamine oxidase (MAO), accepted article 2013, DOI: 10.1111/cbdd.12255.
J. Juárez-Jiménez, E. Mendes, C. Galdeano, C. Martins, D.B. Silva, J. Marco-Contelles, M.C. Carreiras, F.J. Luque, R.R. Ramsay. Exploring the structural basis of the selective inhibition of monoamine oxidase A by dicarbonitrile aminoheterocycles: Role of Asn181 and Ile335 validated by spectroscopic and computational studies. Biochim. Biophys. Acta. accepted article, 2013, doi:pii: S1570-9639(13)00378-6. 10.1016/j.bbapap.2013.11.003
K.I. Shulman, N. Herrmann, S.E. Walker. Current Place of Monoamine Oxidase Inhibitors in the Treatment of Depression. CNS Drugs, 2013, 27, 789-797.
G. Ferino, S. Vilar, M.J. Matos, E. Uriarte, E. Cadoni. Monoamine oxidase inhibitors: Ten years of docking studies. Curr. Top. Med. Chem. 2012, 12, 2145-2162.
K.F. Tipton. Enzymology of monoamine oxidase. Cell. Biochem. Funct. 1986, 4, 79–87.
H.H. Fernandez, J.J. Chen. Monamine oxidase inhibitors: current and emerging agents for Parkinson disease. Clin. Neuropharmacol. 2007, 30, 150–168.
A. Pletscher. The discovery of antidepressants: a winding path. Experientia. 1991, 47, 4–8.
P. Dostert, M.S. Benedetti, K.F. Tipton. Interactions of monoamine oxidase with substrates and inhibitors. Med. Res. Rev. 1989, 9, 45–89.
M.B.H. Youdim, D. Edmondson, K.F. Tipton. The therapeutic potential of monoamine oxidase inhibitors. Nat. Rev. Neurosci. 2006, 7, 295–309.
P. Pacher, V. Kecskemeti. Trends in the development of new antidepressants. Is there a light at the end of the tunnel? Curr Med. Chem. 2004, 11, 925–943.
P. Riederer, L. Lachenmayer, G. Laux. Clinical applications of MAO-inhibitors. Curr. Med. Chem. 2004, 11, 2033–2043.
E.B. Kearney, J.I. Salach, W.H. Walker, R. Seng, T.P. Singer. Structure of the covalently bound flavin of monoamine oxidase. Biochem. Biophys. Res. Commun. 1971, 42, 490–496.
M. Naoi, W. Maruyama. Monoamine oxidase inhibitors as neuroprotective agents in age-dependent neurodegenerative disorders, Curr. Pharmaceut. Des. 2010, 16, 2799-2817.
A. Gaspar, T. Silva, M. Yanez, D. Vina, F. Orallo, F. Ortuso, E. Uriarte, S. Alcaro, F. Borges. Chromone, a privileged scaffold for the development of monoamine oxidase inhibitors. J. Med. Chem. 2011, 54, 5165–5173.
S.M.S. Chauhan, Geetanjali, R. Singh. A mild and efficient synthesis of 10-substituted isoalloxazines in the presence of solid acids. Ind. J. Heterocycl. Chem. 2000, 10, 157-158.
S.M.S. Chauhan, R. Singh, Geetanjali. Microwave assisted synthesis of 10-substituted isoalloxazines in the presence of solid acids. Synth. Commun. 2003, 33, 1179-1184.
Geetanjali, R. Singh. Effect of intramolecular hydrogen bonding on biomimetic reactions by flavins. Heterocyclic Commun. 2013, 19, 261-263.
D.E. Edmondson, A. Mattevi, C. Binda, M. Li, F. Hubálek. Structure and mechanism of monoamine oxidase. Burger's Medicinal Chemistry, Drug Discovery and Development , John Wiley & Sons: New York, 2005.
S.S. Erdem, Ö. Karahan, İ. Yıldız, K. Yelekçi. A computational study on the amine-oxidation mechanism of monoamine oxidase: Insight into the polar nucleophilic mechanism. Org. Biomol. Chem. 2006, 4, 646-658.
J.R. Miller, D.E. Edmondson. Influence of flavin analogue structure on the catalytic activities and flavinylation reactions of recombinant human liver Monoamine Oxidases A and B. J. Biol. Chem. 1999, 274, 23515–23525.
H. Sies. Strategies of antioxidant defense. Eur. J. Biochem. FEBS. 1993, 215, 213-219.
Y. Zhu, P.M. Carvey, Z. Ling. Age-related changes in glutathione and glutathione-related enzymes in rat brain. Brain Res. 2006, 1090, 35-44.
X. Cao, Z. Wei, G.G. Gabriel, X.M. Li, D.D. Mousseau. Calcium- regulation of monoamine oxidase-A contributes to the production of peroxyradicals in hippocampal cultures: implications for Alzheimer disease-related pathology. BMC Neuroscience. 2007, 8, 73-83.
J.C. Shih, K. Chen, M.J. Ridd. Monoamine oxidase: from genes to behavior. Ann. Rev. Neurosc. 1999, 22, 197-217.
S. Mandel, O. Weinreb, T. Amit, M.B. Youdim. Mechanism of neuroprotective action of the anti-Parkinson drug rasagiline and its derivatives. Brain Res. Rev. 2005, 48, 379-387.
J. Saura Marti, R. Kettler, M. Da Prada, J.G. Richards. Molecular neuroanatomy of MAO-A and MAO-B. J. Neural. Transm. Suppl. 1990, 32, 49–53.
O. Weinreb, T. Amit, O. Bar-Am, M.B. Youdim. Rasagiline: a novel anti-Parkinsonian monoamine oxidase-B inhibitor with neuroprotective activity. Prog Neurobiol. 2010, 92, 330–344.
F. Lopez-Munoz, C. Alamo. Monoaminergic neurotransmission: the history of the discovery of antidepressants from 1950s until today. Curr. Pharm. Des. 2009, 15, 1563–1586.
M.P.S. Ishar, G. Singh, S. Singh, K.K. Sreenivasan, G. Singh. Design, synthesis, and evaluation of novel 6-chloro-fluorochromone derivatives as potential topoisomerase inhibitor anticancer agents. Bioorg. Med. Chem. Lett. 2006, 16, 1366–1370.
S. Alcaro, A. Gaspar, F. Ortuso, N. Milhazes, F. Orallo, E. Uriarte, M. Yanez, F. Borges. Chromone-2- and -3-carboxylic acids inhibit differently monoamine oxidases A and B. Bioorg. Med. Chem. Lett. 2010, 20, 2709–2712.
N. Desideri, A. Bolasco, R. Fioravanti, L. Proietti Monaco, F. Orallo, M. Yanez, F. Ortuso, S. Alcaro. Homoisoflavonoids: natural scaffolds with potent and selective monoamine oxidase-B inhibition properties. J. Med. Chem. 2011, 54, 2155–2164.
A. Gaspar, J. Reis, A. Fonseca, N. Milhazes, D. Vina, E. Uriarte, M.F. Borges. Chromone 3-phenylcarboxamides as potent and selective MAO-B inhibitors. Bioorg. Med. Chem. Lett. 2011, 21, 707–709.
P.O. Patil, S.B. Bari, S.D. Firke, P.K. Deshmukh, S.T. Donda, D.A. Patil. A comprehensive review on synthesis and designing aspects of coumarin derivatives as monoamine oxidase inhibitors for depression and Alzheimer’s disease. Bioorg Med Chem. 2013, 21, 2434-2450.
L. Pisani, M. Catto, O. Nicolotti, G. Grossi, M. Di Braccio, R. Soto-Otero, E. Mendez-Alvarez, A. Stefanachi, D. Gadaleta, A. Carotti. Fine molecular tuning at position 4 of 2H-chromen-2-one derivatives in the search of potent and selective monoamine oxidase B inhibitors. Eur. J. Med. Chem. 2013, 70, 723-739.
M. Gerlach, P. Riederer, M.B.H. Youdim. The molecular pharmacology of L-deprenyl. Eur. J. Pharmacol. 1992, 226, 97–108.
K.F. Tipton, S. Boyce, J. O’Sullivan, G.P. Davey, J. Healy. Monoamine Oxidases: Certainties and Uncertainties. Curr. Med. Chem. 2004, 11, 1965-1982.
J.S. Fowler, N.D. Volkow, J. Logan, G.J. Wang, R.R. MacGregor, D. Schlyer, A.P. Wolf, N. Pappas, D. Alexoff, C. Shea, E. Dorflinger, L. Kruchowy, K. Yoo, E. Fazzini, C. Patlak. Slow recovery of human brain MAO B after L-deprenyl (Selegeline) withdrawal. Synaps. 1994, 18, 86-93.
L.J. Legoabe, A. Petzer, J.P. Petzer. Selected chromone derivatives as inhibitors of monoamine oxidase. Bioorg. Med. Chem. Lett. 2012, 22, 5480–5484.
L.J. Legoaba, A. Petzer, J.P. Petzer, Inhibition of monoamine oxidase by selected C6-substituted chromone derivatives. Eur. J. Med. Chem. 2012, 49, 343–353.
C. Binda, J. Wang, L. Pisani, C. Caccia, A. Carotti, P. Salvati, D.E. Edmondson, A. Mattevi, Structures of human monoamine oxidase B complexes with selective noncovalent inhibitors: safinamide and coumarin analogs. J. Med. Chem. 2007, 50, 5848–5852.
L.J. Legoabe, A. Petzer, J.P. Petzer. Selected C7-substituted chromone derivatives as monoamine oxidase inhibitors. Bioorg. Chem. 2012, 45, 1–11.
Y. Zhang, Y. Xu, S. Tan, L. Xu, X. Qian. Rapid and sensitive fluorescent probes for monoamine oxidases B to A at low concentration. Tetrahedron Lett. 2012, 53, 6881-6884.
M.J. Matos, D. Vina, E. Quezada, C. Picciau, G. Delogu, F. Orallo, L. Santana, E. Uriarte. A new series of 3-phenylcoumarins as potent and selective MAO-B inhibitors. Bioorg. Med. Chem. Lett. 2009, 19, 3268-3270.
C. Gnerre, M. Catto, F. Leonetti, P. Weber, P.-A. Carrupt, C. Altomare, A. Carotti, B. Testa. Inhibition of monoamine oxidases by functionalized coumarin derivatives: biological activities, QSARs, and 3D-QSARs. J. Med. Chem. 2000, 43, 4747–4758.
F. Hubálek, C. Binda, A. Khalil, M. Li, A. Mattevi, N. Castagnoli, D.E. Edmondson. Demonstration of Isoleucine 199 as a Structural Determinant for the Selective Inhibition of Human Monoamine Oxidase B by Specific Reversible Inhibitors. J. Biol. Chem. 2005, 280, 15761-15766.
C.I. Manley-King, J.J. Bergh, J.P. Petzer. Inhibition of monoamine oxidase by selected C5- and C6-substituted isatin analogues. Bioorg. Med. Chem. 2011, 19, 261–274.
E.M. Van der Walt, E.M. Milczek, S.F. Malan, D.E. Edmondson, N. Castagnoli Jr, J.J. Bergh, J.P. Petzer. Inhibition of monoamine oxidase by (E)-styrylisatin analogues. Bioorg. Med. Chem. Lett. 2009, 19, 2509-2513.
V Raj. Review on CNS activity of isatin derivatives. Int. J. Curr. Pharmaceut. Res. 2012. 4, 1-9.
A.E. Medvedev, A.S. Ivanov, N.S. Kamyshanskaya, A.Z. Kirkel, T.A. Moskvitina, V.Z. Gorkin, N.Y. Li, VYu, Marshakov. Interaction of indole derivatives with monoamine oxidase A and B. Studies on the structure-inhibitory activity relationship. Biochem. Mol. Biol. Int. 1995, 36, 113-122.
I. Virsis, B.A. Grinberg, D.G. Tsedere, A.A. Prinkulis. Relationship between the structure of 2-indolinones and 2, 3-indolinediones and their biological activity. Pharmaceut. Chem. J. 1985, 19, 554-557.
M.K. Haergreaves, J.G. Pritchard, H.R. Dave. Cyclic carboxylic monoimides. Chem. Res. 1970, 70, 439–469.
C.I. Manley-King, J.J. Bergh, J.P. Petzer. Inhibition of monoamine oxidase by C5-substituted phthalimide analogues. Bioorg. Med. Chem. 2011, 19, 4829–4840.
L. Meiring, J.P. Petzer, A. Petzer. Inhibition of monoamine oxidase by 3,4-dihydro-2(1H)-quinolinone derivatives. Bioorg. Med. Chem. Lett. 2013, 23, 5498–5502.
ISSN 2347–9825
Authors/visitors are advised to use Firefox browser for better experience of journal site.
Open Access: Researcher from developing/low economy countries can access the jorunal contents through WHO-HINARI .
 ISSN 2347-9825
ISSN 2347-9825

